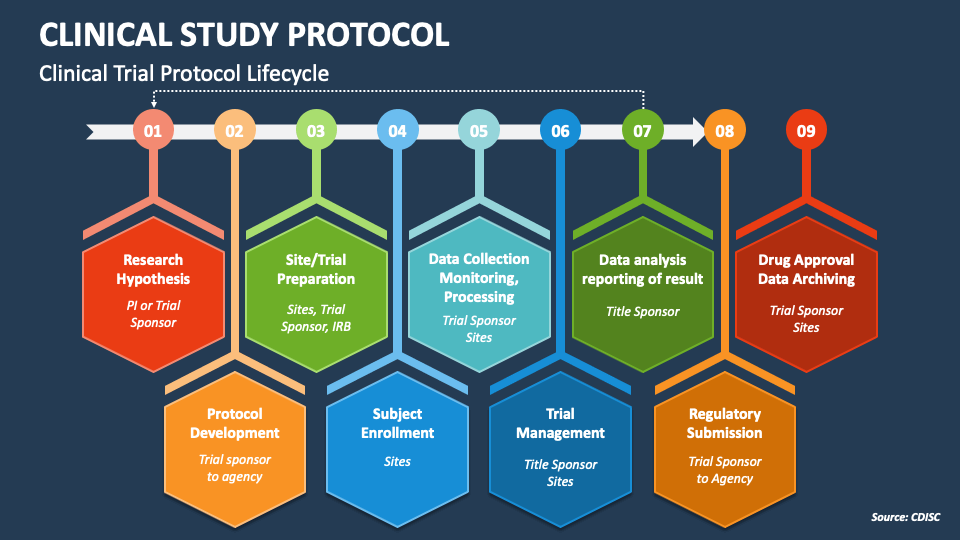
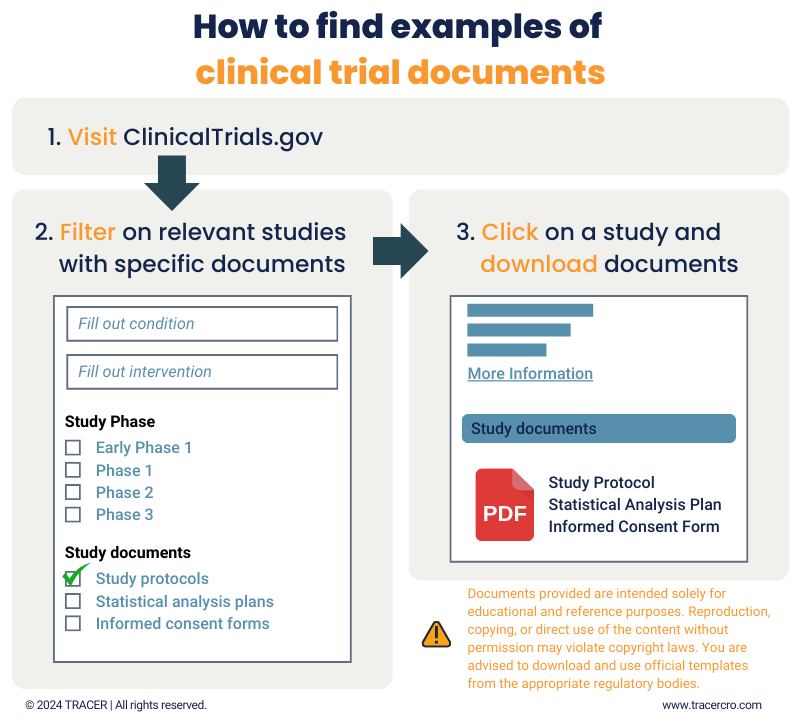
Protocol Template For Clinical Trial - Based on the results of these phase 3 trials, secukinumab received fda approval in january 2016 for the treatment of active psa and as in adult. This preface before finalizing and distributing the clinical trial protocol. The following templates provide a common. Trials is experimenting with a new way of structuring study protocols for randomised trials. This protocol template is a tool to facilitate the development of a research study protocol specifically designed for the investigator initiated. The purpose of this new harmonised guideline is to introduce the clinical protocol template and the technical specification to ensure. This clinical trial protocol template is a suggested format for phase 2 or 3 clinical trials supported by the national institutes of health (nih) that are. The simple innovation is to include all 51 spirit headings and. This template aims to facilitate the development of phase 2 and 3 clinical trial protocols that require a food and drug administration (fda). This clinical trial protocol template is a suggested format for phase 2 and 3.
This template aims to facilitate the development of phase 2 and 3 clinical trial protocols that require a food and drug administration (fda). This protocol template is a tool to facilitate the development of a research study protocol specifically designed for the investigator initiated. The simple innovation is to include all 51 spirit headings and. This clinical trial protocol template is a suggested format for phase 2 or 3 clinical trials supported by the national institutes of health (nih) that are. The purpose of this new harmonised guideline is to introduce the clinical protocol template and the technical specification to ensure. This preface before finalizing and distributing the clinical trial protocol. Based on the results of these phase 3 trials, secukinumab received fda approval in january 2016 for the treatment of active psa and as in adult. This clinical trial protocol template is a suggested format for phase 2 and 3. Trials is experimenting with a new way of structuring study protocols for randomised trials. The following templates provide a common.
This clinical trial protocol template is a suggested format for phase 2 or 3 clinical trials supported by the national institutes of health (nih) that are. The purpose of this new harmonised guideline is to introduce the clinical protocol template and the technical specification to ensure. This template aims to facilitate the development of phase 2 and 3 clinical trial protocols that require a food and drug administration (fda). This preface before finalizing and distributing the clinical trial protocol. The following templates provide a common. This protocol template is a tool to facilitate the development of a research study protocol specifically designed for the investigator initiated. Based on the results of these phase 3 trials, secukinumab received fda approval in january 2016 for the treatment of active psa and as in adult. This clinical trial protocol template is a suggested format for phase 2 and 3. Trials is experimenting with a new way of structuring study protocols for randomised trials. The simple innovation is to include all 51 spirit headings and.
Free Clinical Trial Templates Smartsheet
This clinical trial protocol template is a suggested format for phase 2 and 3. This protocol template is a tool to facilitate the development of a research study protocol specifically designed for the investigator initiated. This template aims to facilitate the development of phase 2 and 3 clinical trial protocols that require a food and drug administration (fda). Based on.
Clinical Trial Protocol Synopsis Template
The simple innovation is to include all 51 spirit headings and. This preface before finalizing and distributing the clinical trial protocol. This template aims to facilitate the development of phase 2 and 3 clinical trial protocols that require a food and drug administration (fda). This protocol template is a tool to facilitate the development of a research study protocol specifically.
Phase 1 Clinical Trial Protocol Template
This preface before finalizing and distributing the clinical trial protocol. The following templates provide a common. This clinical trial protocol template is a suggested format for phase 2 or 3 clinical trials supported by the national institutes of health (nih) that are. This clinical trial protocol template is a suggested format for phase 2 and 3. The purpose of this.
Clinical Study Protocol (CSP) Template Clinical Study Templates
Based on the results of these phase 3 trials, secukinumab received fda approval in january 2016 for the treatment of active psa and as in adult. This clinical trial protocol template is a suggested format for phase 2 and 3. This clinical trial protocol template is a suggested format for phase 2 or 3 clinical trials supported by the national.
Fillable Online clinicaltrials.govProvidedDocs66CIRB Clinical Trial
This protocol template is a tool to facilitate the development of a research study protocol specifically designed for the investigator initiated. The following templates provide a common. Based on the results of these phase 3 trials, secukinumab received fda approval in january 2016 for the treatment of active psa and as in adult. This preface before finalizing and distributing the.
Clinical Study Protocol PowerPoint and Google Slides Template PPT Slides
This template aims to facilitate the development of phase 2 and 3 clinical trial protocols that require a food and drug administration (fda). The following templates provide a common. Trials is experimenting with a new way of structuring study protocols for randomised trials. This protocol template is a tool to facilitate the development of a research study protocol specifically designed.
Free Doctor Visit Summary Template to Edit Online
Trials is experimenting with a new way of structuring study protocols for randomised trials. This template aims to facilitate the development of phase 2 and 3 clinical trial protocols that require a food and drug administration (fda). The following templates provide a common. This clinical trial protocol template is a suggested format for phase 2 and 3. This protocol template.
Fillable Online Clinical Trial Protocol leemsProtocol Templates for
The purpose of this new harmonised guideline is to introduce the clinical protocol template and the technical specification to ensure. Based on the results of these phase 3 trials, secukinumab received fda approval in january 2016 for the treatment of active psa and as in adult. This clinical trial protocol template is a suggested format for phase 2 and 3..
Clinical trial protocol template and example to download TRACER
The simple innovation is to include all 51 spirit headings and. This clinical trial protocol template is a suggested format for phase 2 and 3. Trials is experimenting with a new way of structuring study protocols for randomised trials. This template aims to facilitate the development of phase 2 and 3 clinical trial protocols that require a food and drug.
Phase 1 Clinical Trial Protocol Template
Based on the results of these phase 3 trials, secukinumab received fda approval in january 2016 for the treatment of active psa and as in adult. This clinical trial protocol template is a suggested format for phase 2 and 3. This protocol template is a tool to facilitate the development of a research study protocol specifically designed for the investigator.
This Protocol Template Is A Tool To Facilitate The Development Of A Research Study Protocol Specifically Designed For The Investigator Initiated.
This template aims to facilitate the development of phase 2 and 3 clinical trial protocols that require a food and drug administration (fda). Based on the results of these phase 3 trials, secukinumab received fda approval in january 2016 for the treatment of active psa and as in adult. The following templates provide a common. The simple innovation is to include all 51 spirit headings and.
This Preface Before Finalizing And Distributing The Clinical Trial Protocol.
This clinical trial protocol template is a suggested format for phase 2 and 3. Trials is experimenting with a new way of structuring study protocols for randomised trials. The purpose of this new harmonised guideline is to introduce the clinical protocol template and the technical specification to ensure. This clinical trial protocol template is a suggested format for phase 2 or 3 clinical trials supported by the national institutes of health (nih) that are.