Mdsap Audit Report Template - It describes the interim process for auditing organizations to submit mdsap audit report packages to the regulatory authorities. These documents set expectations on auditing organizations (aos) on how to meet the requirements of the program. A list of resources for the medical device single audit program (mdsap). Home medical devices cdrh international affairs medical device single audit program (mdsap) mdsap policies, procedures, templates and forms Mdsap databases identifier in preparation for audits and assessment 20 may 2025 form mdsap as f0010.1 Explore mdsap audit procedures and forms for harmonized regulatory audits of medical device manufacturers across multiple jurisdictions.
Mdsap databases identifier in preparation for audits and assessment 20 may 2025 form mdsap as f0010.1 These documents set expectations on auditing organizations (aos) on how to meet the requirements of the program. Explore mdsap audit procedures and forms for harmonized regulatory audits of medical device manufacturers across multiple jurisdictions. Home medical devices cdrh international affairs medical device single audit program (mdsap) mdsap policies, procedures, templates and forms It describes the interim process for auditing organizations to submit mdsap audit report packages to the regulatory authorities. A list of resources for the medical device single audit program (mdsap).
Mdsap databases identifier in preparation for audits and assessment 20 may 2025 form mdsap as f0010.1 Explore mdsap audit procedures and forms for harmonized regulatory audits of medical device manufacturers across multiple jurisdictions. Home medical devices cdrh international affairs medical device single audit program (mdsap) mdsap policies, procedures, templates and forms These documents set expectations on auditing organizations (aos) on how to meet the requirements of the program. It describes the interim process for auditing organizations to submit mdsap audit report packages to the regulatory authorities. A list of resources for the medical device single audit program (mdsap).
MDSAP Audit Complete Guide! Elexes
Mdsap databases identifier in preparation for audits and assessment 20 may 2025 form mdsap as f0010.1 A list of resources for the medical device single audit program (mdsap). Home medical devices cdrh international affairs medical device single audit program (mdsap) mdsap policies, procedures, templates and forms It describes the interim process for auditing organizations to submit mdsap audit report packages.
MDSAP Audit Process & Sequence Oriel STAT A MATRIX
Home medical devices cdrh international affairs medical device single audit program (mdsap) mdsap policies, procedures, templates and forms Mdsap databases identifier in preparation for audits and assessment 20 may 2025 form mdsap as f0010.1 A list of resources for the medical device single audit program (mdsap). Explore mdsap audit procedures and forms for harmonized regulatory audits of medical device manufacturers.
Fillable Online MDSAP Audit Procedures and FormsFDA Fax Email Print
These documents set expectations on auditing organizations (aos) on how to meet the requirements of the program. Mdsap databases identifier in preparation for audits and assessment 20 may 2025 form mdsap as f0010.1 A list of resources for the medical device single audit program (mdsap). Home medical devices cdrh international affairs medical device single audit program (mdsap) mdsap policies, procedures,.
MDSAP Audit Procedures and s Doc Template pdfFiller
Explore mdsap audit procedures and forms for harmonized regulatory audits of medical device manufacturers across multiple jurisdictions. Home medical devices cdrh international affairs medical device single audit program (mdsap) mdsap policies, procedures, templates and forms A list of resources for the medical device single audit program (mdsap). These documents set expectations on auditing organizations (aos) on how to meet the.
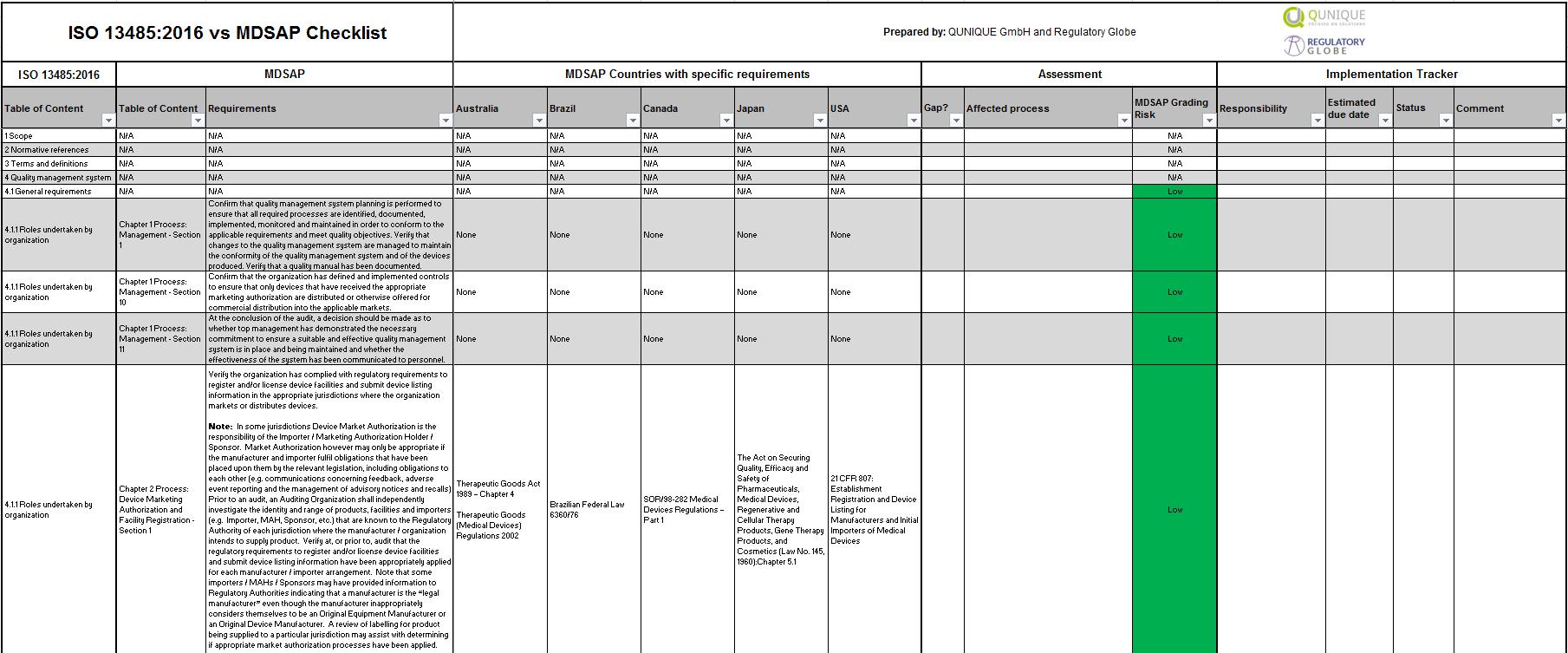
Free MDSAP Audit Checklist PDF SafetyCulture
Mdsap databases identifier in preparation for audits and assessment 20 may 2025 form mdsap as f0010.1 It describes the interim process for auditing organizations to submit mdsap audit report packages to the regulatory authorities. A list of resources for the medical device single audit program (mdsap). These documents set expectations on auditing organizations (aos) on how to meet the requirements.
MDSAP Medical Device Single Audit Program (Ultimate Guide)
Mdsap databases identifier in preparation for audits and assessment 20 may 2025 form mdsap as f0010.1 It describes the interim process for auditing organizations to submit mdsap audit report packages to the regulatory authorities. A list of resources for the medical device single audit program (mdsap). These documents set expectations on auditing organizations (aos) on how to meet the requirements.
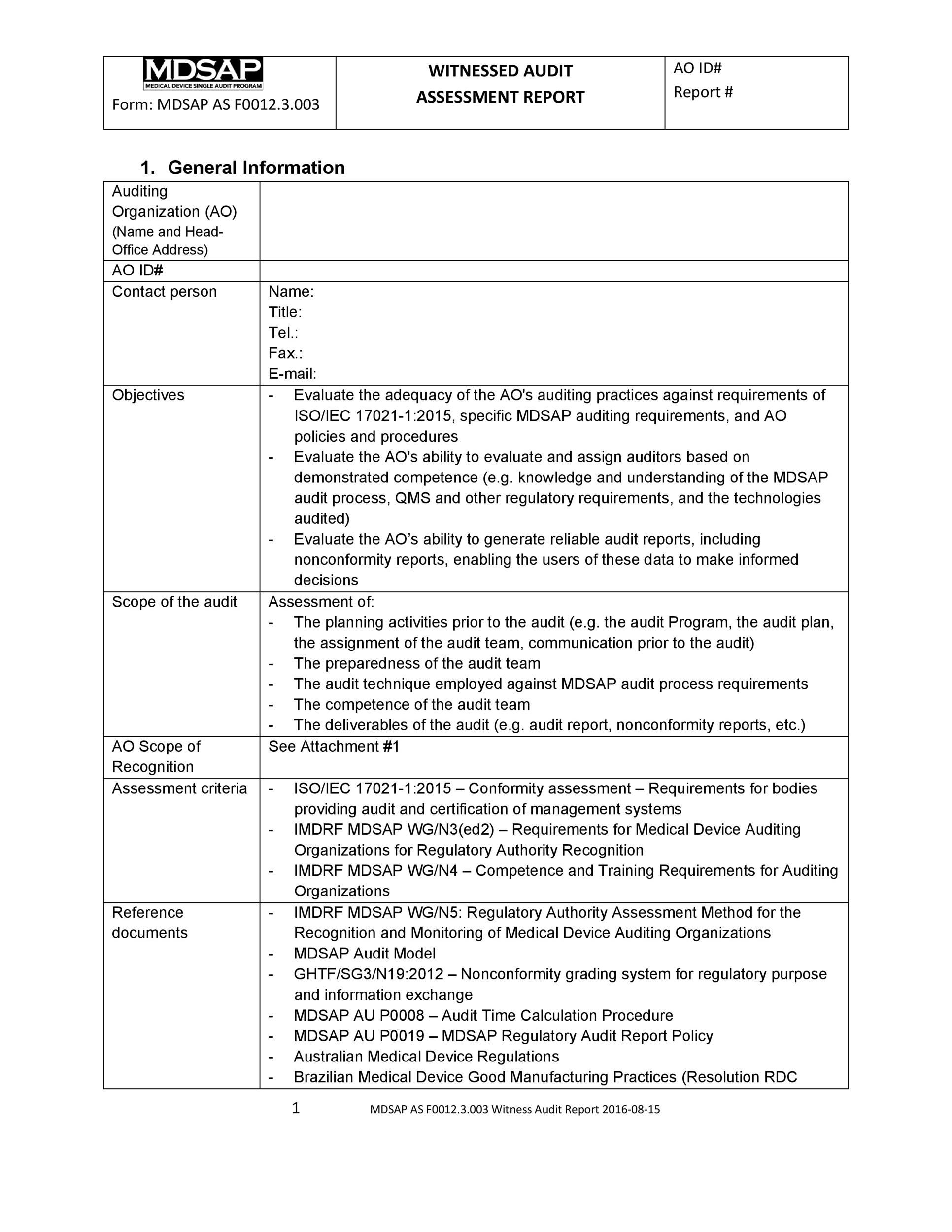
50 Free Audit Report Templates (Internal Audit Reports) ᐅ TemplateLab
Home medical devices cdrh international affairs medical device single audit program (mdsap) mdsap policies, procedures, templates and forms These documents set expectations on auditing organizations (aos) on how to meet the requirements of the program. Explore mdsap audit procedures and forms for harmonized regulatory audits of medical device manufacturers across multiple jurisdictions. A list of resources for the medical device.
MDSAP Medical Device Single Audit Program Advanxa
A list of resources for the medical device single audit program (mdsap). Home medical devices cdrh international affairs medical device single audit program (mdsap) mdsap policies, procedures, templates and forms Mdsap databases identifier in preparation for audits and assessment 20 may 2025 form mdsap as f0010.1 These documents set expectations on auditing organizations (aos) on how to meet the requirements.
Mdsap Audit Model USA Wholesale brunofuga.adv.br
These documents set expectations on auditing organizations (aos) on how to meet the requirements of the program. It describes the interim process for auditing organizations to submit mdsap audit report packages to the regulatory authorities. Home medical devices cdrh international affairs medical device single audit program (mdsap) mdsap policies, procedures, templates and forms Explore mdsap audit procedures and forms for.
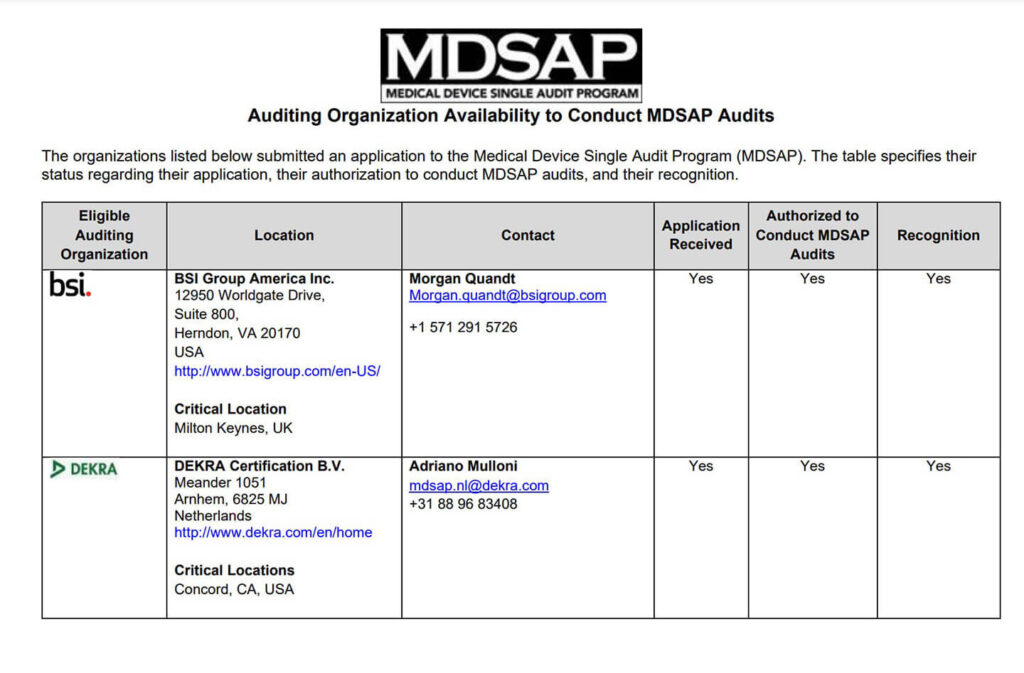
MDSAP Regulatory Globe
A list of resources for the medical device single audit program (mdsap). These documents set expectations on auditing organizations (aos) on how to meet the requirements of the program. Home medical devices cdrh international affairs medical device single audit program (mdsap) mdsap policies, procedures, templates and forms Mdsap databases identifier in preparation for audits and assessment 20 may 2025 form.
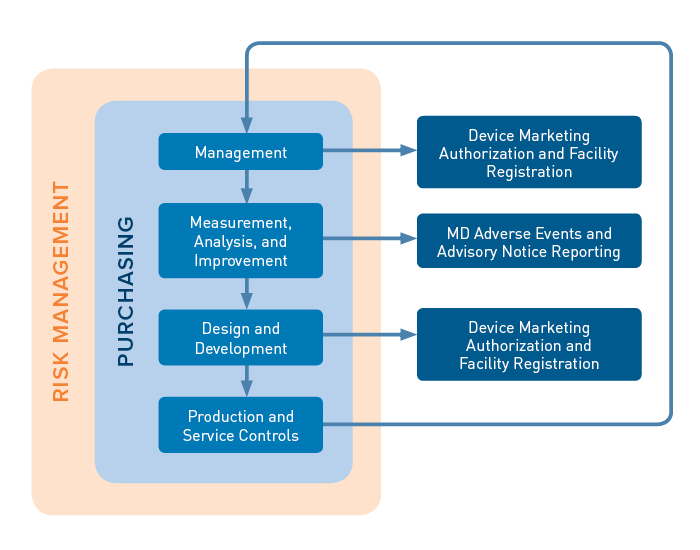
Explore Mdsap Audit Procedures And Forms For Harmonized Regulatory Audits Of Medical Device Manufacturers Across Multiple Jurisdictions.
These documents set expectations on auditing organizations (aos) on how to meet the requirements of the program. It describes the interim process for auditing organizations to submit mdsap audit report packages to the regulatory authorities. Mdsap databases identifier in preparation for audits and assessment 20 may 2025 form mdsap as f0010.1 Home medical devices cdrh international affairs medical device single audit program (mdsap) mdsap policies, procedures, templates and forms