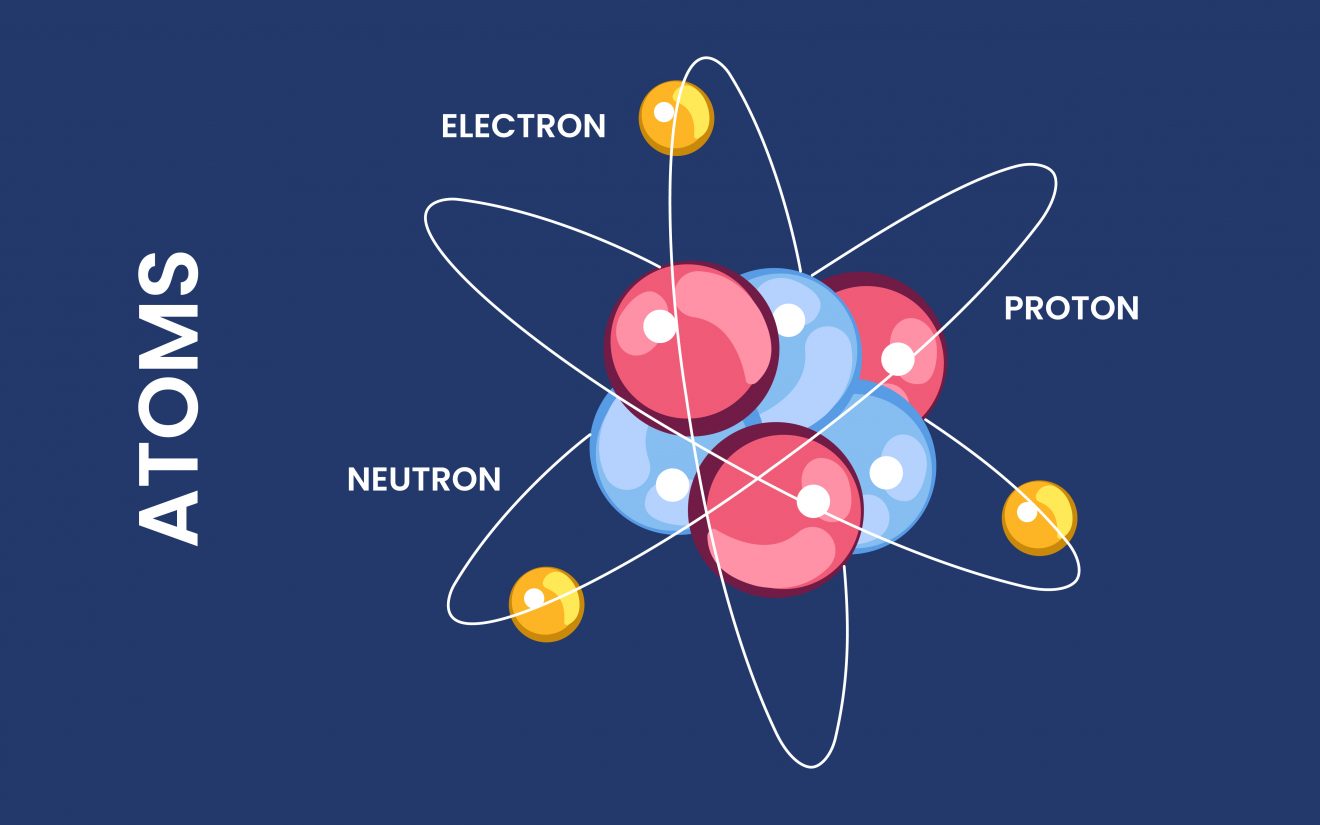
How Are Atoms Formed - Atoms can be combined with other atoms to form molecules, but they cannot. Dalton stated that all atoms of an element are identical in shape, size, and mass. Democritus believed that atoms were uniform, solid, hard, incompressible, and indestructible and that they moved in infinite. The tiny particles called atoms are the basic building blocks of all matter. However, atoms are made up of three types of subatomic particles: Elements are characterized by the mass of their atoms. The heavy protons and neutrons that make up the nucleus (the central. In 1808 the english chemist john dalton suggested that each element consists of identical atoms, and in 1811 the italian physicist amedeo avogadro.
Dalton stated that all atoms of an element are identical in shape, size, and mass. Atoms can be combined with other atoms to form molecules, but they cannot. However, atoms are made up of three types of subatomic particles: Elements are characterized by the mass of their atoms. In 1808 the english chemist john dalton suggested that each element consists of identical atoms, and in 1811 the italian physicist amedeo avogadro. The heavy protons and neutrons that make up the nucleus (the central. Democritus believed that atoms were uniform, solid, hard, incompressible, and indestructible and that they moved in infinite. The tiny particles called atoms are the basic building blocks of all matter.
Elements are characterized by the mass of their atoms. Democritus believed that atoms were uniform, solid, hard, incompressible, and indestructible and that they moved in infinite. In 1808 the english chemist john dalton suggested that each element consists of identical atoms, and in 1811 the italian physicist amedeo avogadro. The heavy protons and neutrons that make up the nucleus (the central. Atoms can be combined with other atoms to form molecules, but they cannot. However, atoms are made up of three types of subatomic particles: The tiny particles called atoms are the basic building blocks of all matter. Dalton stated that all atoms of an element are identical in shape, size, and mass.
Atomic Structures & the Periodic Table VISTA HEIGHTS 8TH GRADE SCIENCE
Atoms can be combined with other atoms to form molecules, but they cannot. Dalton stated that all atoms of an element are identical in shape, size, and mass. Democritus believed that atoms were uniform, solid, hard, incompressible, and indestructible and that they moved in infinite. The heavy protons and neutrons that make up the nucleus (the central. However, atoms are.
The Structure of Atoms
In 1808 the english chemist john dalton suggested that each element consists of identical atoms, and in 1811 the italian physicist amedeo avogadro. Atoms can be combined with other atoms to form molecules, but they cannot. However, atoms are made up of three types of subatomic particles: Dalton stated that all atoms of an element are identical in shape, size,.
Atom Definition, Structure, & Parts with Labeled Diagram
Democritus believed that atoms were uniform, solid, hard, incompressible, and indestructible and that they moved in infinite. In 1808 the english chemist john dalton suggested that each element consists of identical atoms, and in 1811 the italian physicist amedeo avogadro. Dalton stated that all atoms of an element are identical in shape, size, and mass. Atoms can be combined with.
What is Atom? How does it Exist? and it's Symbols Teachoo
Democritus believed that atoms were uniform, solid, hard, incompressible, and indestructible and that they moved in infinite. Dalton stated that all atoms of an element are identical in shape, size, and mass. The heavy protons and neutrons that make up the nucleus (the central. However, atoms are made up of three types of subatomic particles: Atoms can be combined with.
Basic Structure of Atom Learn Definition, Facts and Examples
In 1808 the english chemist john dalton suggested that each element consists of identical atoms, and in 1811 the italian physicist amedeo avogadro. However, atoms are made up of three types of subatomic particles: Dalton stated that all atoms of an element are identical in shape, size, and mass. Democritus believed that atoms were uniform, solid, hard, incompressible, and indestructible.
The Atom. ppt download
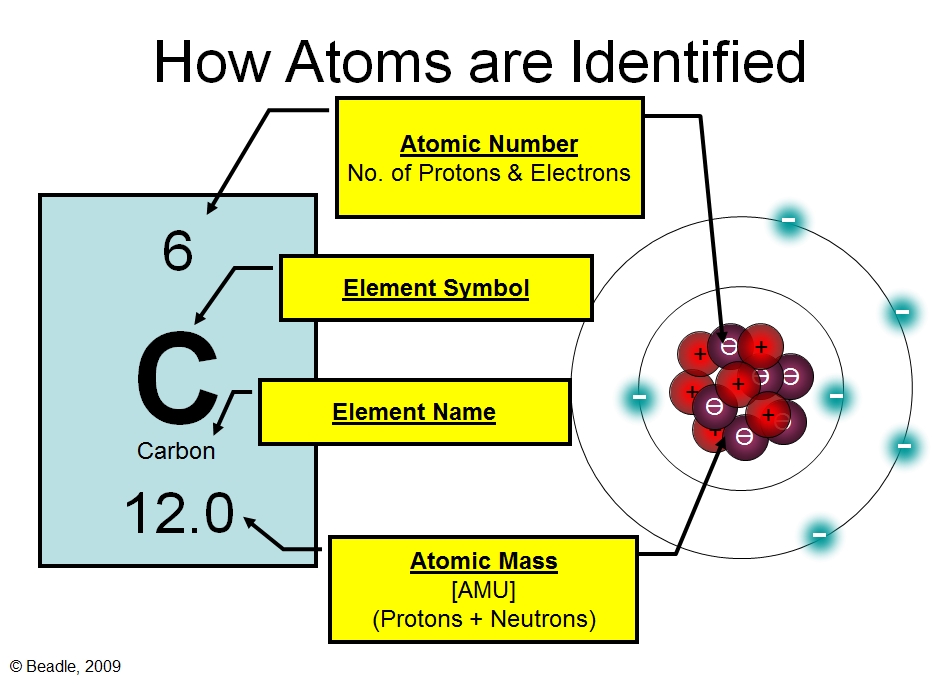
Elements are characterized by the mass of their atoms. Dalton stated that all atoms of an element are identical in shape, size, and mass. The tiny particles called atoms are the basic building blocks of all matter. However, atoms are made up of three types of subatomic particles: In 1808 the english chemist john dalton suggested that each element consists.
Learn the Parts of an Atom
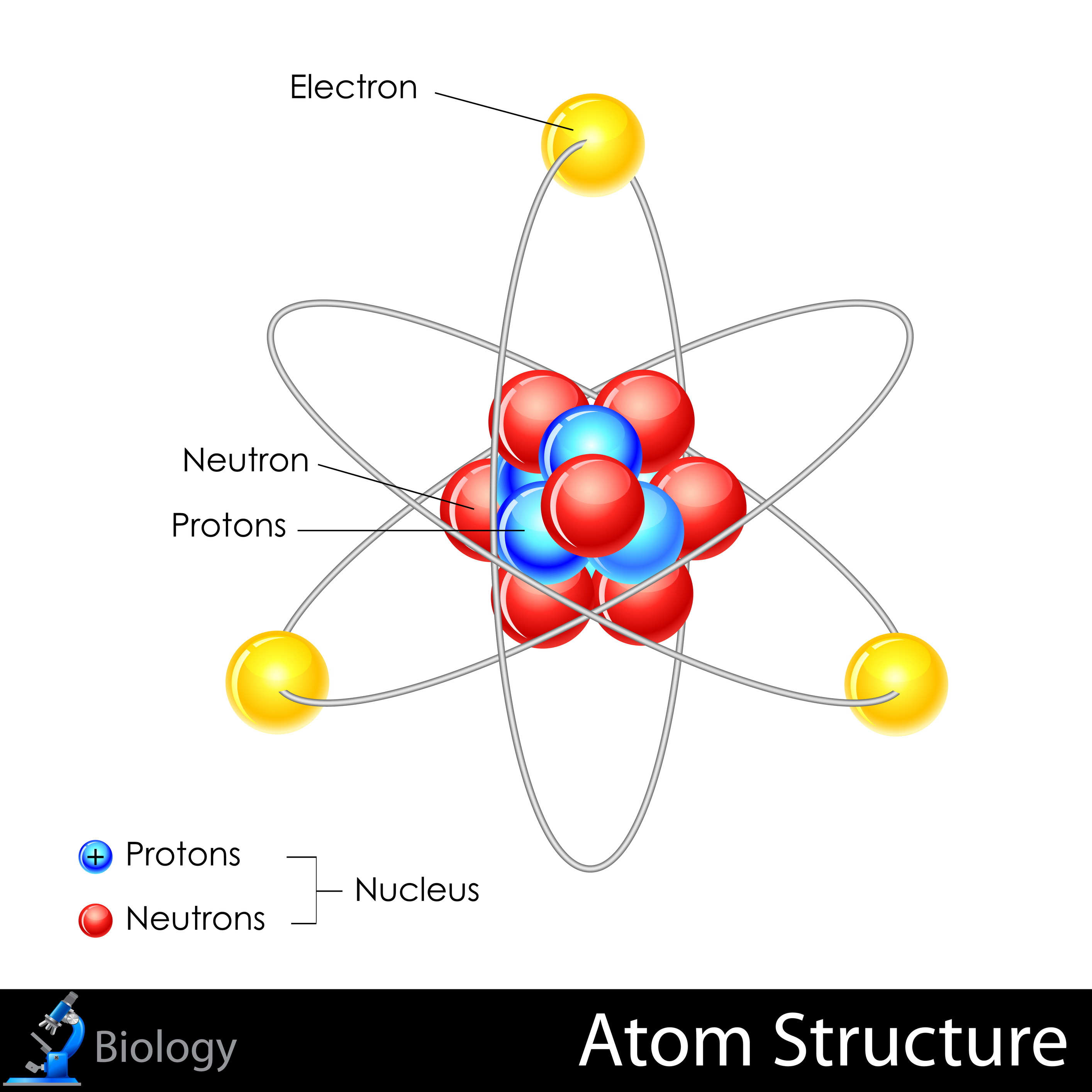
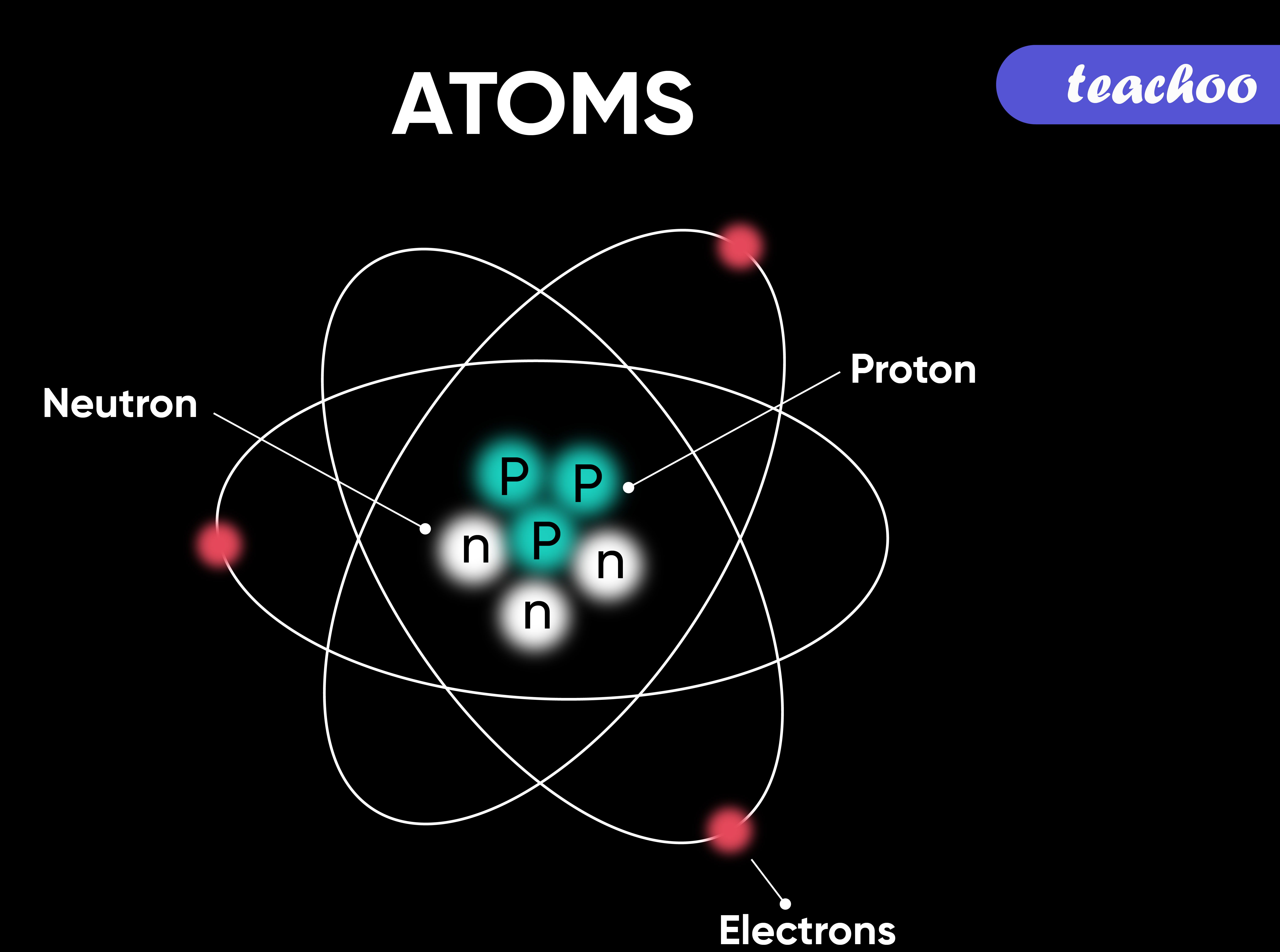
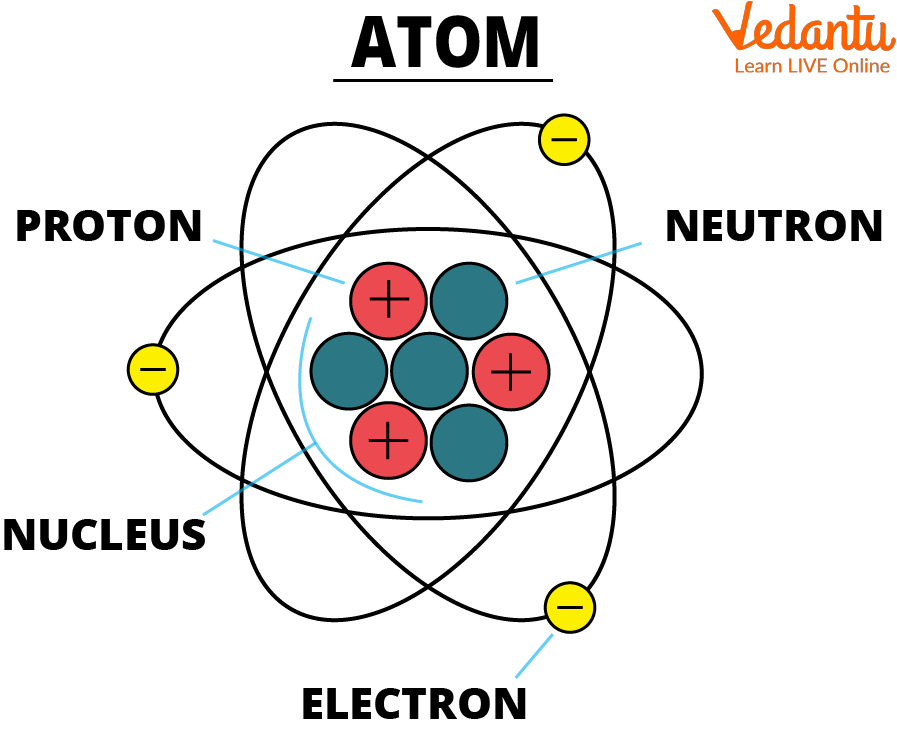
However, atoms are made up of three types of subatomic particles: The tiny particles called atoms are the basic building blocks of all matter. Elements are characterized by the mass of their atoms. Democritus believed that atoms were uniform, solid, hard, incompressible, and indestructible and that they moved in infinite. The heavy protons and neutrons that make up the nucleus.
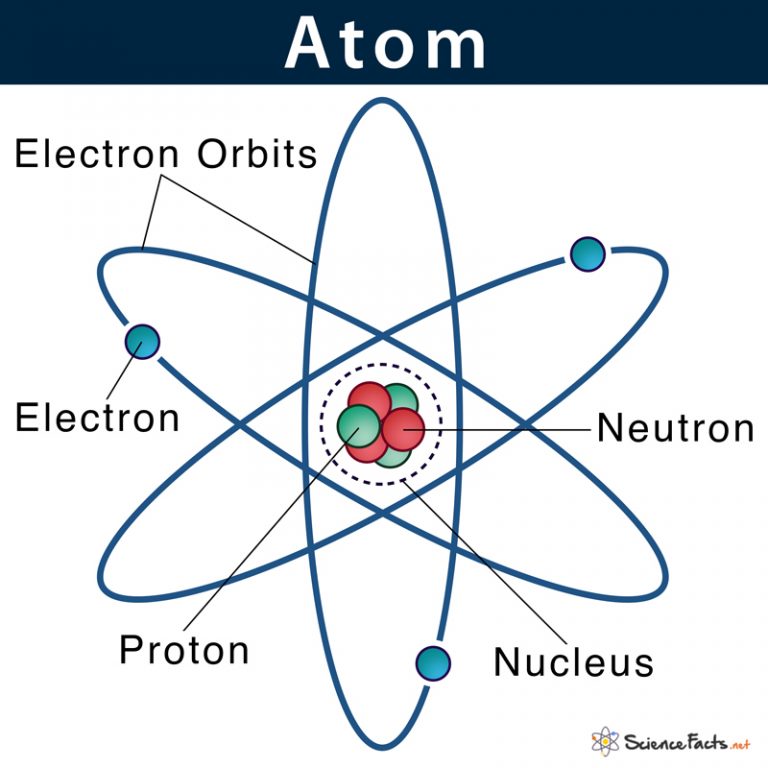
Basic Model of the Atom Atomic Theory
In 1808 the english chemist john dalton suggested that each element consists of identical atoms, and in 1811 the italian physicist amedeo avogadro. Elements are characterized by the mass of their atoms. The heavy protons and neutrons that make up the nucleus (the central. Atoms can be combined with other atoms to form molecules, but they cannot. The tiny particles.
Atom Facts 15 Fascinating Tidbits to Explore the Microscopic World
Dalton stated that all atoms of an element are identical in shape, size, and mass. In 1808 the english chemist john dalton suggested that each element consists of identical atoms, and in 1811 the italian physicist amedeo avogadro. The heavy protons and neutrons that make up the nucleus (the central. Elements are characterized by the mass of their atoms. However,.
Diagram of atom structure illustration Stock Vector Image & Art Alamy
Dalton stated that all atoms of an element are identical in shape, size, and mass. Democritus believed that atoms were uniform, solid, hard, incompressible, and indestructible and that they moved in infinite. In 1808 the english chemist john dalton suggested that each element consists of identical atoms, and in 1811 the italian physicist amedeo avogadro. The tiny particles called atoms.
Dalton Stated That All Atoms Of An Element Are Identical In Shape, Size, And Mass.
However, atoms are made up of three types of subatomic particles: Elements are characterized by the mass of their atoms. The tiny particles called atoms are the basic building blocks of all matter. Democritus believed that atoms were uniform, solid, hard, incompressible, and indestructible and that they moved in infinite.
In 1808 The English Chemist John Dalton Suggested That Each Element Consists Of Identical Atoms, And In 1811 The Italian Physicist Amedeo Avogadro.
The heavy protons and neutrons that make up the nucleus (the central. Atoms can be combined with other atoms to form molecules, but they cannot.


/GettyImages-141483984-56a133b65f9b58b7d0bcfdb1.jpg)