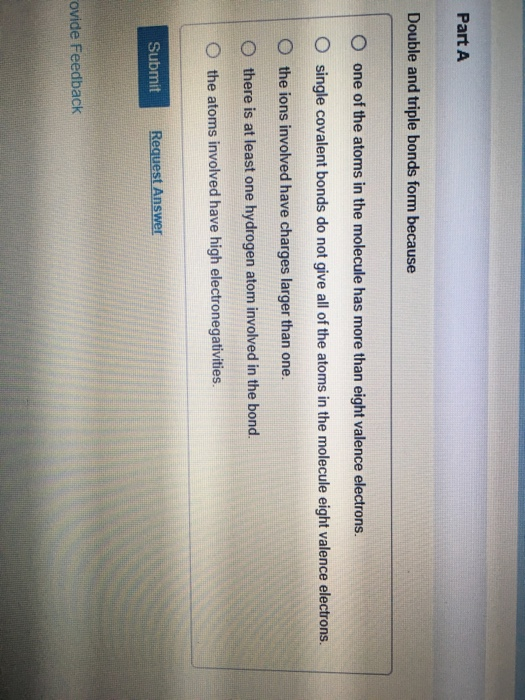
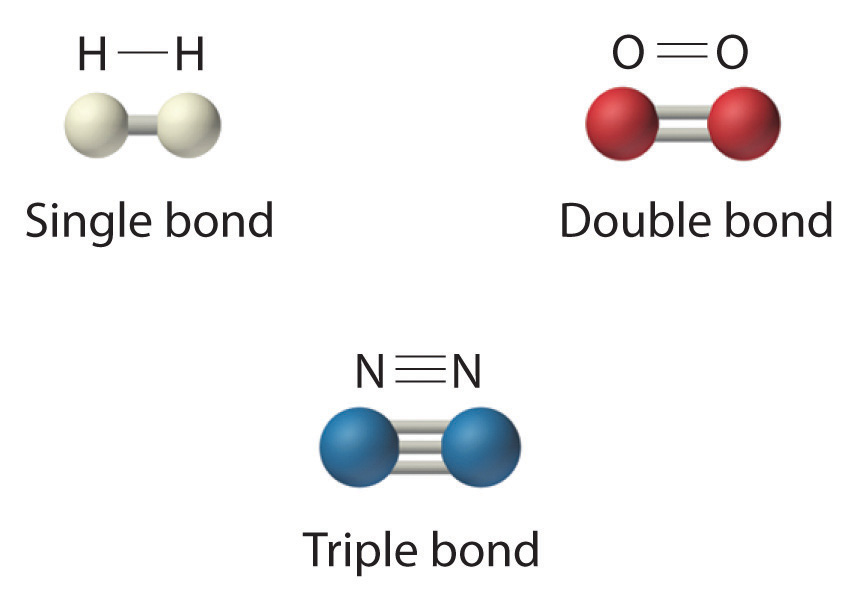
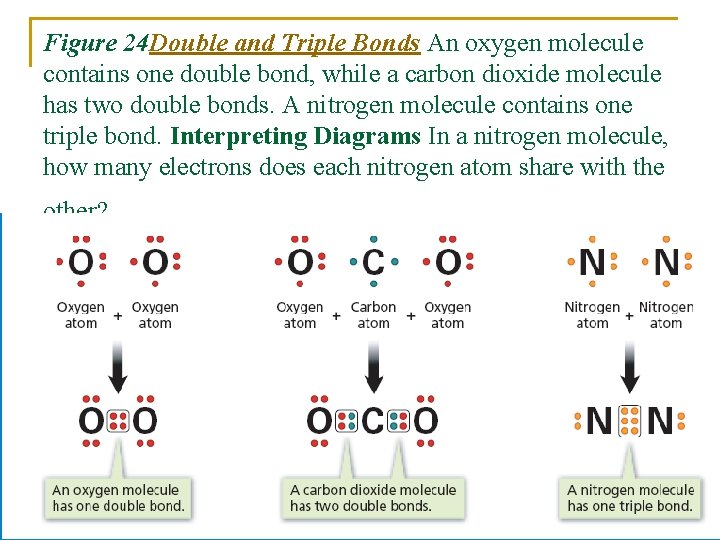
Double And Triple Bonds Form Because - B) single covalent bonds do not give all of the. Single covalent bonds do not give all of the atoms in the molecule eight. Double and triple bonds form primarily because of option b: The atoms involved have high electronegativities b. Question 8 double and triple bonds form because a. Unsaturated fatty acids are the molecules which is an example of a carbon chain that is bent because of the carbon's ability to. One of the atoms in the molecule has more than. They allow atoms to share more than one pair of electrons, increasing stability. Double and triple bonds form because: A) the atoms involved have high electronegativities.
They allow atoms to share more than one pair of electrons, increasing stability. Double and triple bonds form because: Double and triple bonds form primarily because of option b: B) single covalent bonds do not give all of the. Double and triple bonds form because: One of the atoms in the molecule has more than. Unsaturated fatty acids are the molecules which is an example of a carbon chain that is bent because of the carbon's ability to. The atoms involved have high electronegativities b. Question 8 double and triple bonds form because a. Single covalent bonds do not give all of the atoms in the molecule eight.
Unsaturated fatty acids are the molecules which is an example of a carbon chain that is bent because of the carbon's ability to. The atoms involved have high electronegativities b. B) single covalent bonds do not give all of the. A) the atoms involved have high electronegativities. Double and triple bonds form primarily because of option b: Single covalent bonds do not give all of the atoms in the molecule eight. They allow atoms to share more than one pair of electrons, increasing stability. Double and triple bonds form because: Double and triple bonds form because: Question 8 double and triple bonds form because a.
Chemical Bonding. ppt download
Double and triple bonds form primarily because of option b: Question 8 double and triple bonds form because a. Single covalent bonds do not give all of the atoms in the molecule eight. Double and triple bonds form because: Unsaturated fatty acids are the molecules which is an example of a carbon chain that is bent because of the carbon's.
Solved Part A Double and triple bonds form because O one of
B) single covalent bonds do not give all of the. Single covalent bonds do not give all of the atoms in the molecule eight. The atoms involved have high electronegativities b. A) the atoms involved have high electronegativities. Question 8 double and triple bonds form because a.
Hybridization Dr. Harris Lecture 11 (Ch ) 9/25/12 ppt video online
Double and triple bonds form primarily because of option b: B) single covalent bonds do not give all of the. Unsaturated fatty acids are the molecules which is an example of a carbon chain that is bent because of the carbon's ability to. Question 8 double and triple bonds form because a. The atoms involved have high electronegativities b.
PPT Covalent Bonding PowerPoint Presentation, free download ID2844390
Double and triple bonds form because: B) single covalent bonds do not give all of the. They allow atoms to share more than one pair of electrons, increasing stability. Single covalent bonds do not give all of the atoms in the molecule eight. A) the atoms involved have high electronegativities.
Chemical Compounds
B) single covalent bonds do not give all of the. They allow atoms to share more than one pair of electrons, increasing stability. A) the atoms involved have high electronegativities. Single covalent bonds do not give all of the atoms in the molecule eight. Unsaturated fatty acids are the molecules which is an example of a carbon chain that is.
LECTURE 3.4 LEWIS STRUCTURES ppt download
They allow atoms to share more than one pair of electrons, increasing stability. A) the atoms involved have high electronegativities. Double and triple bonds form because: B) single covalent bonds do not give all of the. Double and triple bonds form primarily because of option b:
Covalent Bonding Part 1 Lewis Structures ppt download
Unsaturated fatty acids are the molecules which is an example of a carbon chain that is bent because of the carbon's ability to. Question 8 double and triple bonds form because a. They allow atoms to share more than one pair of electrons, increasing stability. One of the atoms in the molecule has more than. The atoms involved have high.
Chpt. 5 Chemical Bonding Chemical Formulas ppt download
Double and triple bonds form because: Double and triple bonds form primarily because of option b: Question 8 double and triple bonds form because a. Single covalent bonds do not give all of the atoms in the molecule eight. B) single covalent bonds do not give all of the.
Covalent Bonds How Covalent Bonds Form n n
Single covalent bonds do not give all of the atoms in the molecule eight. They allow atoms to share more than one pair of electrons, increasing stability. A) the atoms involved have high electronegativities. B) single covalent bonds do not give all of the. Double and triple bonds form because:
Chapter Atomic Structure and Chemical Bonds ppt download
Unsaturated fatty acids are the molecules which is an example of a carbon chain that is bent because of the carbon's ability to. Single covalent bonds do not give all of the atoms in the molecule eight. Double and triple bonds form because: A) the atoms involved have high electronegativities. One of the atoms in the molecule has more than.
Single Covalent Bonds Do Not Give All Of The Atoms In The Molecule Eight.
Question 8 double and triple bonds form because a. Double and triple bonds form primarily because of option b: B) single covalent bonds do not give all of the. They allow atoms to share more than one pair of electrons, increasing stability.
Unsaturated Fatty Acids Are The Molecules Which Is An Example Of A Carbon Chain That Is Bent Because Of The Carbon's Ability To.
A) the atoms involved have high electronegativities. The atoms involved have high electronegativities b. Double and triple bonds form because: Double and triple bonds form because: