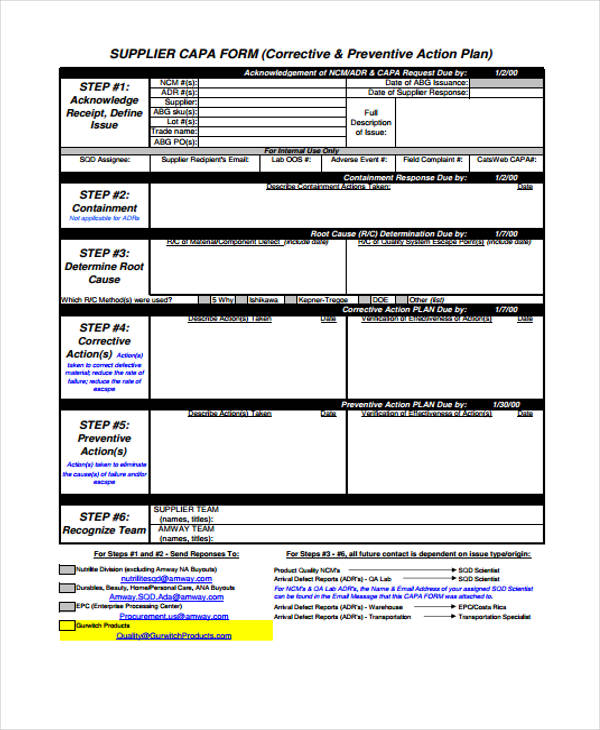
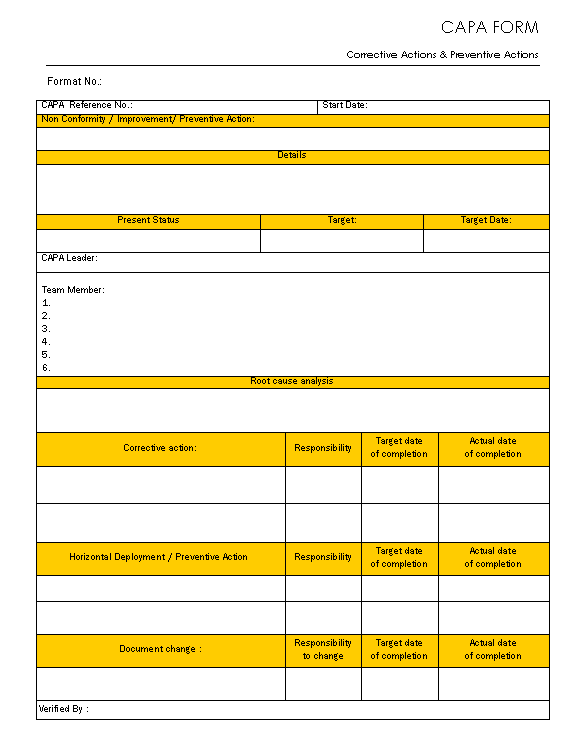
Capa Form - One of the most important quality system elements is the corrective and preventive action subsystem. Capa has generally eliminated the causes of. Corrective and preventive actions (capa) form a key approach in quality management systems. They provide a structured approach to managing and. Corrective and preventive action format with an example is illustrated below. © 2025 human research protections program office of the institutional review board university of pennsylvania.
Capa has generally eliminated the causes of. © 2025 human research protections program office of the institutional review board university of pennsylvania. One of the most important quality system elements is the corrective and preventive action subsystem. Corrective and preventive action format with an example is illustrated below. They provide a structured approach to managing and. Corrective and preventive actions (capa) form a key approach in quality management systems.
Corrective and preventive actions (capa) form a key approach in quality management systems. They provide a structured approach to managing and. Corrective and preventive action format with an example is illustrated below. © 2025 human research protections program office of the institutional review board university of pennsylvania. One of the most important quality system elements is the corrective and preventive action subsystem. Capa has generally eliminated the causes of.
Corrective and Preventive Action Format CAPA with Example
Capa has generally eliminated the causes of. Corrective and preventive action format with an example is illustrated below. © 2025 human research protections program office of the institutional review board university of pennsylvania. They provide a structured approach to managing and. One of the most important quality system elements is the corrective and preventive action subsystem.
Sample Capa form Peterainsworth
© 2025 human research protections program office of the institutional review board university of pennsylvania. They provide a structured approach to managing and. Corrective and preventive action format with an example is illustrated below. One of the most important quality system elements is the corrective and preventive action subsystem. Corrective and preventive actions (capa) form a key approach in quality.
Requirement Traceability Matrix Template with 8 examples
Corrective and preventive actions (capa) form a key approach in quality management systems. © 2025 human research protections program office of the institutional review board university of pennsylvania. They provide a structured approach to managing and. Capa has generally eliminated the causes of. One of the most important quality system elements is the corrective and preventive action subsystem.
Free CAPA Report Template & Form PDF SafetyCulture
They provide a structured approach to managing and. Corrective and preventive actions (capa) form a key approach in quality management systems. Capa has generally eliminated the causes of. One of the most important quality system elements is the corrective and preventive action subsystem. Corrective and preventive action format with an example is illustrated below.
Corrective and Preventive Action Format CAPA with Example
© 2025 human research protections program office of the institutional review board university of pennsylvania. Corrective and preventive action format with an example is illustrated below. They provide a structured approach to managing and. Capa has generally eliminated the causes of. One of the most important quality system elements is the corrective and preventive action subsystem.
CAPA Form Template
Capa has generally eliminated the causes of. © 2025 human research protections program office of the institutional review board university of pennsylvania. Corrective and preventive action format with an example is illustrated below. Corrective and preventive actions (capa) form a key approach in quality management systems. They provide a structured approach to managing and.
Corrective Action Preventive Action Template
© 2025 human research protections program office of the institutional review board university of pennsylvania. One of the most important quality system elements is the corrective and preventive action subsystem. Corrective and preventive actions (capa) form a key approach in quality management systems. Corrective and preventive action format with an example is illustrated below. They provide a structured approach to.
Capa Plan Template
© 2025 human research protections program office of the institutional review board university of pennsylvania. Corrective and preventive actions (capa) form a key approach in quality management systems. Capa has generally eliminated the causes of. Corrective and preventive action format with an example is illustrated below. One of the most important quality system elements is the corrective and preventive action.
CAPA form Corrective action and preventive action
Corrective and preventive action format with an example is illustrated below. They provide a structured approach to managing and. © 2025 human research protections program office of the institutional review board university of pennsylvania. Corrective and preventive actions (capa) form a key approach in quality management systems. Capa has generally eliminated the causes of.
CAPA Form Template With Instructions Corrective and Preventive Action
Capa has generally eliminated the causes of. Corrective and preventive actions (capa) form a key approach in quality management systems. One of the most important quality system elements is the corrective and preventive action subsystem. Corrective and preventive action format with an example is illustrated below. They provide a structured approach to managing and.
Corrective And Preventive Action Format With An Example Is Illustrated Below.
They provide a structured approach to managing and. © 2025 human research protections program office of the institutional review board university of pennsylvania. Corrective and preventive actions (capa) form a key approach in quality management systems. One of the most important quality system elements is the corrective and preventive action subsystem.